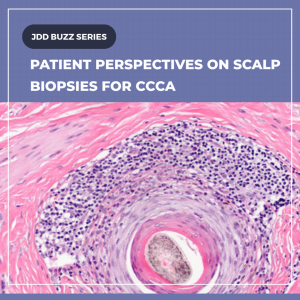
A recent Journal of Drugs in Dermatology brief communication and interview with Temitayo A. Ogunleye, MD, explore why patients often decline scalp biopsy despite its role in securing a diagnosis that can enable hair‑saving treatment. Key insights: Patients who consented demonstrated clearer understanding and greater trust in their clinicians; those who declined frequently had knowledge gaps rather than firm opposition, and many remained open to biopsy if their condition progressed.
Practical takeaways:
- Introduce biopsy early and link it to outcomes patients value
- Use clear, patient‑centered explanations about risks, benefits, and how biopsy may change management
- Proactively address common concerns and build trust
- Revisit the conversation over time—many patients will reconsider as the disease evolves
For dermatologists treating CCCA in skin of color, prioritizing education and shared decision‑making can improve diagnostic yield and treatment outcomes.